Hello,
Welcome to Insider Healthcare. I'm healthcare editor Leah Rosenbaum, and this week in healthcare news:
- Insider went to the HLTH conference in Boston where they spoke with execs at Google and other companies;
- The FDA and CDC recommended booster shots of the Moderna and Johnson & Johnson COVID-19 vaccine;
- A "mix-and-match" strategy of COVID-19 booster shots could be even more effective at creating immunity.
If you're new to this newsletter, sign up here. Comments, tips? Email me at [email protected] or tweet @leah_rosenbaum. Let's get to it…

Dr. Karen DeSalvo, Google's chief health officer.
Google Health
At the HLTH 2021 conference, execs spoke about the future of healthcare

Google Health
This week, we sent almost all of our healthcare team out to Boston for the 2021 HLTH conference.
Our reporters met with dozens of executives to find out what the next big trends in healthcare will be.
- We spoke onstage with Google's health chief about what's next for the company after it recently dissolved its health division.
- Shelby Livingston tuned into what Best Buy Health's president about the electronics retailer's foray into healthcare.
- Andrew Dunn quizzed Regeneron's top scientist about the company's new genetic research.
- And Jade Khatib reported on how healthcare providers are meeting underserved patients online in an effort to quell misinformation.
We'll have more coverage of HLTH in the weeks ahead. In the meantime, we want to know, what did you take away from the conference? Reply to Blake Dodge's tweet asking that here.
Read more>>
Google's health chief lays out the company's next steps after shying away from a full healthcare business
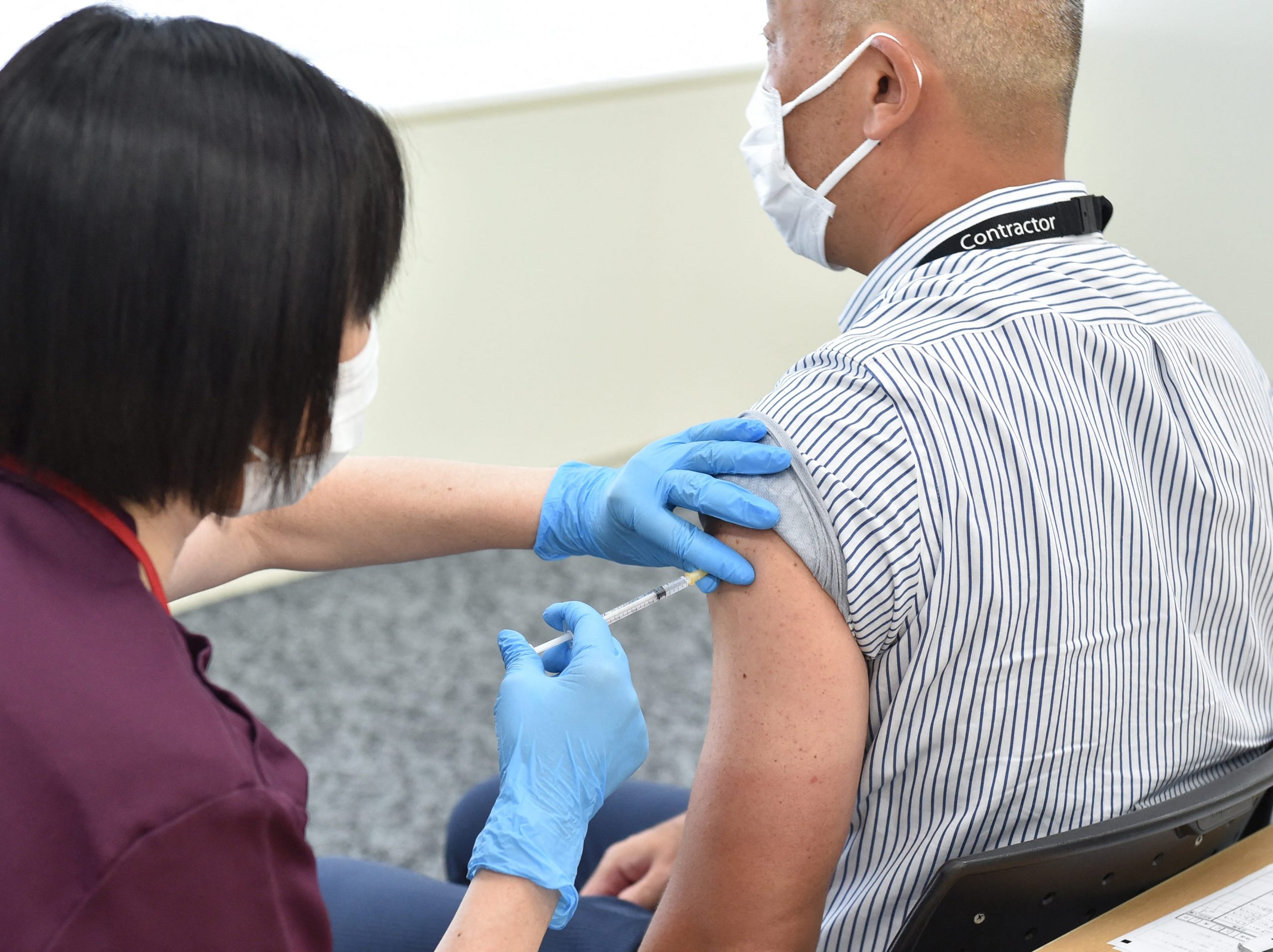
An employee (R) of Japan's Suntory Holdings receives the Moderna coronavirus vaccine for Covid-19 .
KAZUHIRO NOGI/AFP via Getty Images
FDA and CDC OK booster shots for Moderna and Johnson & Johnson COVID-19 vaccines

KAZUHIRO NOGI/AFP via Getty Images
In an exciting announcement on Wednesday, the FDA authorized booster shots for Moderna's COVID-19 vaccine to people at high risk for severe disease and older adults. People in these categories can get booster shots 6 months after their second Moderna shot.
At the same time, the FDA authorized a second dose of the Johnson & Johnson COVID-19 vaccine for anyone who got a single dose of the vaccine at least 2 months ago. Andrew and I covered the big news.
On Thursday, the CDC agreed with the FDA and booster shots became official US policy.
Up next: an expert FDA panel is set on Tuesday to review the evidence around the use of Pfizer's vaccine in kids 5-11. Ahead of that recommendation, Pfizer released new results on Friday that show the vaccine is 91% effective in that age group.
Here's what you need to know>>
The FDA just authorized booster shots of Moderna's and Johnson & Johnson's COVID-19 vaccines and is letting users mix and match shots
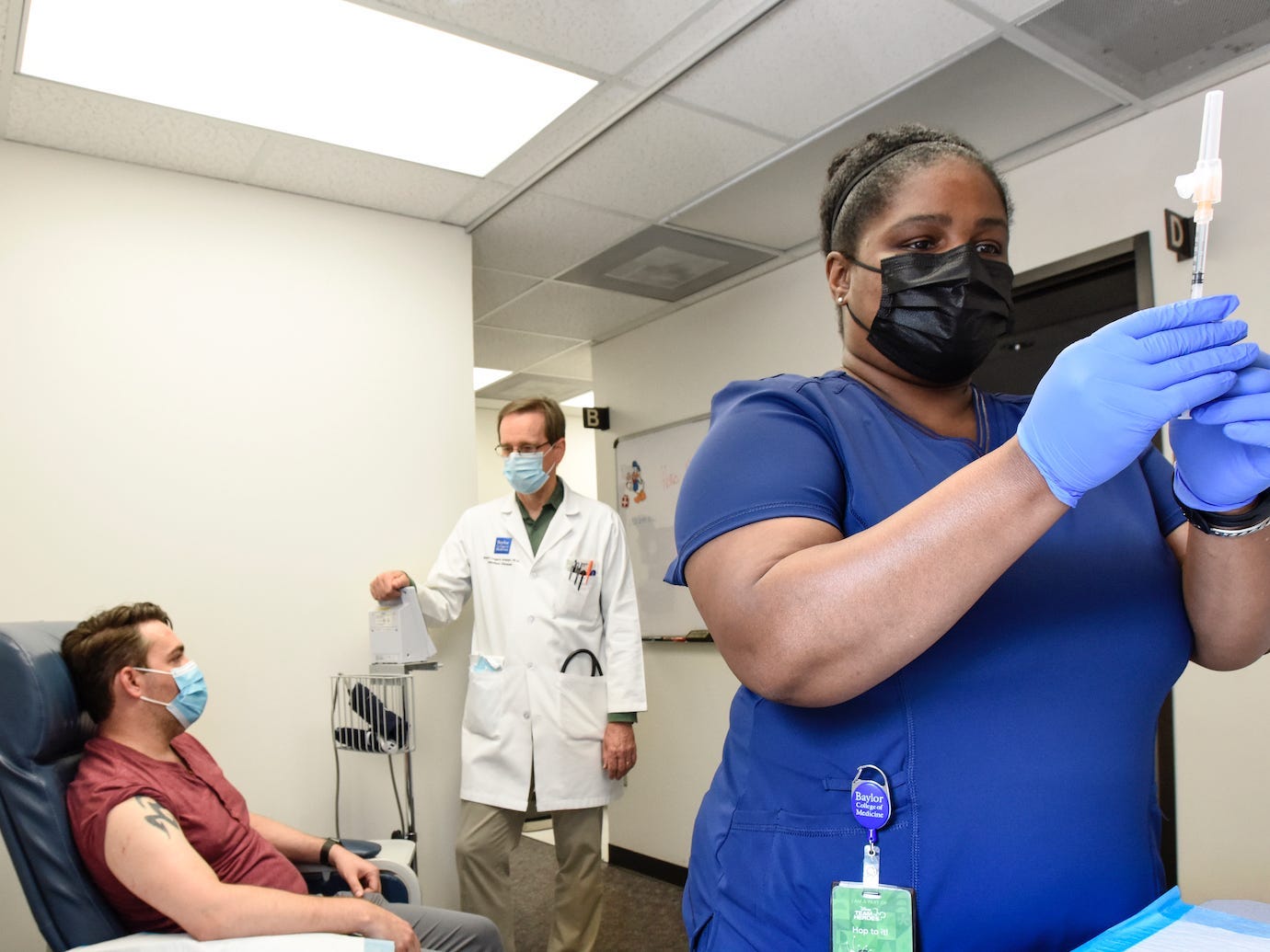
Chanei Henry, senior research coordinator of molecular virology and microbiology at Baylor College of Medicine, prepares a COVID-19 vaccine.
Baylor College of Medicine
Mix & match becomes mainstream

Baylor College of Medicine
Researchers have been talking for months about the potential benefit to "mixing-and-matching" COVID-19 vaccines and booster shots.
On Wednesday, the FDA announced that the agency now authorizes people to do it. These "heterologous" vaccines can sometimes produce a stronger immune response, research has found.
Dr. Catherine Schuster-Bruce and Hilary Brueck discuss how they work.
Dive in>>
How 'mix and match' COVID-19 booster shots work, and why we're using them
More stories that kept us busy this week:
- Megan Hernbroth got the pitchdeck that a dermatology company used to raise money from Livongo's founder Glen Tullman.
- Allison DeAngelis wrote about some experts that are skeptical of the use of blood-based cancer tests.
- Venture capital correspondent April Joyner wrote about a new acquisition by home testing kit company Everlywell.
- Burger icon In-N-Out is getting in trouble for not checking vaccine cards, according to business reporter Kate Duffy.
- Science senior reporter Aria Bendix wrote about when the right time is to get your seasonal flu shot.
-Leah
